Around 50 percent of people harbour yeast fungi in their bodies. In general, our immune systems manage to keep these troublesome tenants under control. However, yeast fungi do cause several thousand deaths each year in Germany alone – and the figures are rising. People with weakened immune systems, such as chemotherapy patients and transplant recipients, are particularly susceptible to potentially fatal mycoses, which are aggressive fungal infections. At present, only a few drugs are available for the treatment of such diseases and some of them can cause considerable side-effects. To make matters worse, the fungi are becoming increasingly resistant to the drugs used to combat them. Now, researchers from Fraunhofer IGB and experts from EMC microcollections GmbH are collaborating on a research project aimed at identifying new compounds which have a broad spectrum of effect against a variety of different fungi and are more easily tolerated than conventional antimycotics.
Novel screening assay for antimycotics
Screening for antimycotics

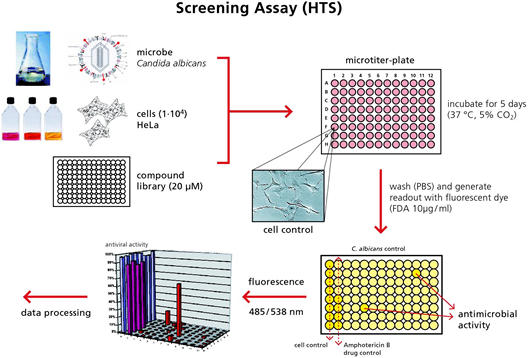
The screening for novel antimycotics is performed on the basis of a substance library, which consists of thousands of potential active substance compounds produced by EMC microcollections GmbH via microchemical synthesis. We at Fraunhofer IGB then pre-select potential antimycotic compounds by means of chemical structure. In the following antimycotic screening we test these compounds to assess their tolerability and effectiveness using our newly developed assay system (Figure 1). These special testing regimes allow us to investigate, together with further partners from antiinfectives screening, the effects of the test substances on growth of the yeast fungus Candida albicans in the presence of human cells (HeLa cell line). Thus, we can ensure that only compounds which kill fungi but are well-tolerated by the human body go forward for further testing [1].
First hits already identified
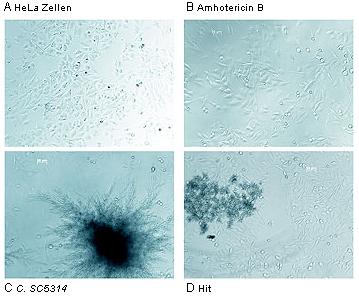
Initial results from the screening are already available. We have identified several hits, promising individual compounds which merit further investigation. Some of these compounds are currently being modified chemically to improve their effectiveness even further and to analyze their structure-effect relationship. In a second investigation phase, the focus will on identifying the molecular mode of effect of the hit substances and their further development into pharmaceutically interesting active substances for more effective treatment of fungal infections.
References
[1] Gerald Kleymann & Hans-Otto Werling (2004) A generally applicable, high-throughput screening-compatible assay to identify, evaluate and optimise antimicrobial agents for drug therapy, Journal of Biomolecular Screening (JBS) 9(7): 578-587
 Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB
Fraunhofer Institute for Interfacial Engineering and Biotechnology IGB